Analysis of Benzoin by SFC-PDA-CD Method Scouting System – 10 columns x 3 solvents for rapid screening
April 15, 2026Introduction
Supercritical fluid chromatography (SFC) employs carbon dioxide as the primary component of the mobile phase and is characterized by its ability to maintain high separation efficiency even at high flow rates (linear velocities). This capability makes SFC particularly well suited for the rapid separation and fractionation of chiral compounds. To determine optimal separation conditions, method scouting approaches are commonly used, in which analytes are systematically screened across a range of modifier solvents and chiral stationary phases. In recent years, chiral columns packed with 3 µm particles have been developed, offering improved efficiency and significantly faster separations compared to conventional 5 µm particle size columns traditionally used in SFC.
The circular dichroism (CD) detector used in this study provides simultaneous acquisition of UV chromatograms, CD chromatograms, and the g-factor (CD/UV ratio) in real time. In CD chromatograms, optical isomers produce peaks with opposite signs, enabling unambiguous identification of enantiomers even when the elution order is reversed during method screening.
In this application note, rapid method scouting for the chiral separation of benzoin*1 was performed using a JASCO SFC system equipped with the Method Scouting Assistant Program, an add-in software package for ChromNAV Ver. 2. Benzoin, an aromatic compound, was evaluated using three modifier solvents and ten chiral columns with a 3 µm particles, enabling high-speed and high-efficiency screening.
*1 Benzoin here refers to the chemical compound, not the fragrance material of the same name.
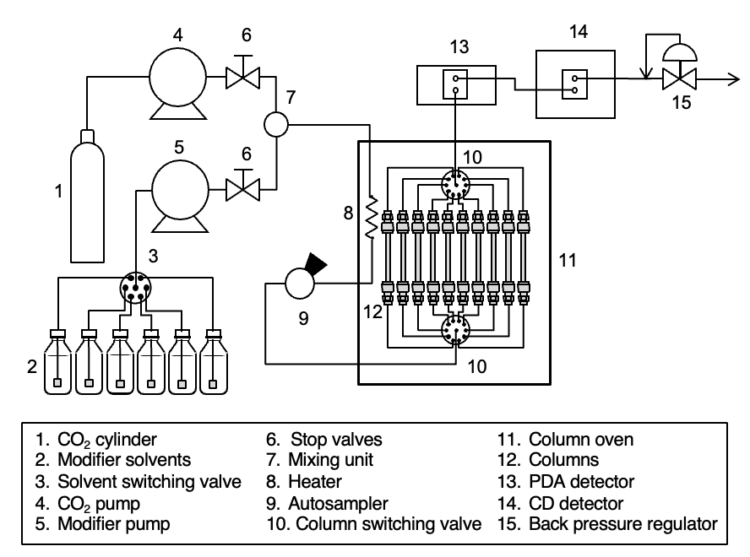
Experimental
Instruments
CO2 pump: PU-4380
Modifier pump: PU-4185*
Autosampler: AS-4350
Column oven: CO-4065*
PDA detector: MD-4010*
CD detector: CD-4095*
BP regulator: BP-4340
*with option units
SFC Conditions
Column: CHIRALPAK IA/SFC, IB-N/SFC, IC/SFC, ID/SFC, IE/SFC, IF/SFC, IG/SFC, IH/SFC, IJ/SFC, IK/SFC
(3.0 mm I.D. x 50 mm L, 3 µm)*
Eluent: Carbon dioxide/modifier (75/25)
Modifier: A; methanol, B; acetonitrile/ethanol (80/20), C; t-butyl methyl ether/ethanol (80/20)
Flow rate: 1.2 mL/min
Column temp: 40 ºC
Wavelength: 230 nm (MD–4010), 230 nm (CD–4095)
Back pressure: 10 MPa
Injection volume: 1 µL
Sample: 0.5 mg/mL benzoin in methanol
*CHIRALPAK is a trademark or registered mark of Daicel Corporation.
Keywords
Benzoin, supercritical fluid chromatography, SFC, method screening, method scouting, chiral separation, circular dichroism detector, CD detector, CHIRALPAK, 3 µm
Results
During method scouting, the column stabilization time was set to 10 minutes for the first column following solvent changeover and 3 minutes for subsequent columns to allow sufficient equilibration. The analysis time for each run was set to 3.0 minutes, resulting in a total screening time of approximately 3.4 hours per compound.
Figure 3–1 shows the screening results obtained using a photodiode array (PDA) detector at a wavelength of 230 nm, while Figure 3–2 shows the corresponding results obtained using a circular dichroism (CD) detector at a wavelength of 230 nm. In these figures, each row represents a different modifier solvent, and each column represents a different chiral column. In the CD chromatograms, enantiomers produce signals of opposite sign, enabling clear and immediate differentiation of optical isomers.
Table 1 summarizes the resolution for hydroxyzine under each condition. Results with a resolution of 1.0 or less were classified as incomplete separation (I.S.). Among the conditions evaluated, several combinations achieved complete separation across all modifier systems. Notably, the use of t-butyl methyl ether/ethanol (80/20) as the modifier generally resulted in increased retention and improved resolution compared to the other modifiers tested.
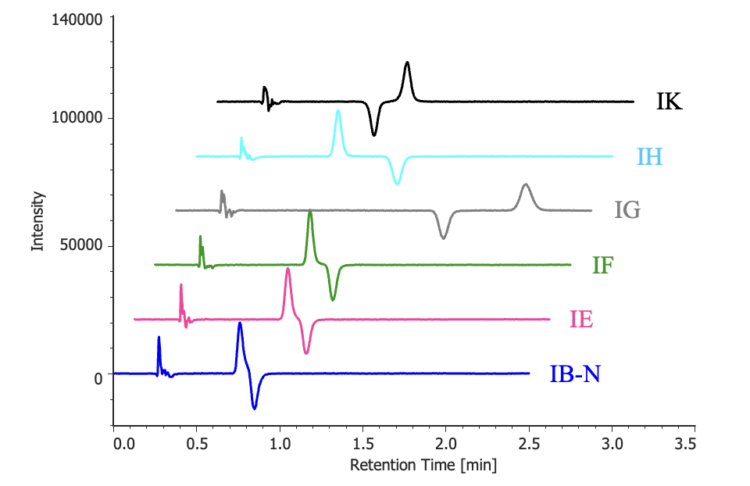
Figure 4 shows the CD chromatograms obtained for columns that achieved chiral separation under the t-butyl methyl ether/ethanol (80/20) conditions. Analysis of the CD data confirmed that the IG and IK columns exhibited a reversed elution order relative to the other columns.
Conclusion
In this application note, the use of 3 µm particle size chiral columns in SFC enabled rapid and efficient method scouting, allowing optimal chiral separation conditions for benzoin to be identified within approximately 3.4 hours. Conditions that yielded partial or near-baseline separation may be further optimized through adjustments to column dimensions, modifier composition or additive concentration to achieve baseline separation.
Monitoring CD chromatograms obtained with the circular dichroism detector enabled clear identification of elution order reversals arising from differences in chiral stationary phases.
Compared to conventional HPLC, SFC offers significantly faster method development as well as reduced solvent consumption. The SFC method scouting system used in this application note provides a powerful and efficient approach for the rapid screening and optimization of chiral separations, making it highly valuable for both analytical and preparative applications.
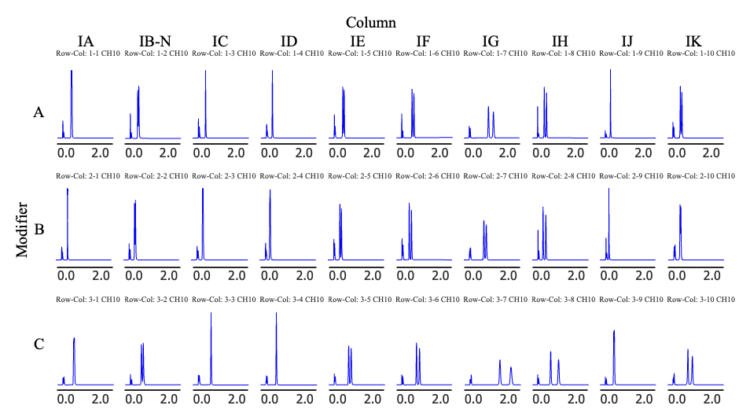
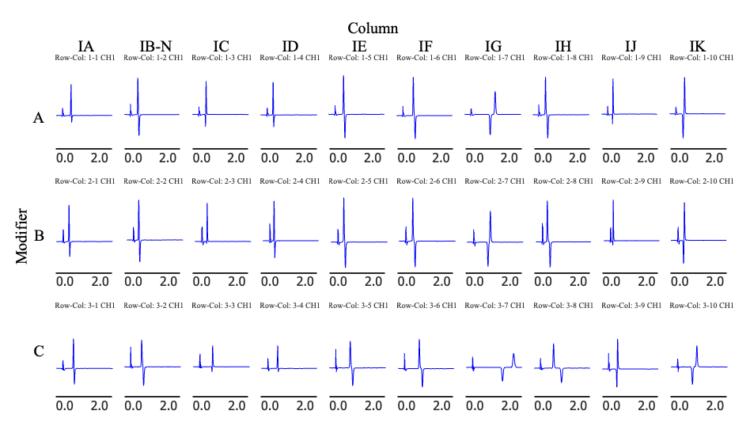
Table 1. Comparison of resolution
| Modifier / Column | IA | IB-N | IC | ID | IE | IF | IG | IH | IJ | IK |
|---|---|---|---|---|---|---|---|---|---|---|
| Methanol | N.S. | 1.12 | N.S. | N.S. | 1.39 | 1.85 | 3.00 | 2.33 | N.S. | 1.69 |
| Acetonitrile / Ethanol | N.S. | 1.09 | N.S. | N.S. | 1.62 | 2.39 | 1.82 | 2.73 | N.S. | I.S. |
| t-Butyl methyl ether / Ethanol | I.S. | 1.65 | N.S. | N.S. | 1.77 | 2.38 | 4.50 | 4.38 | I.S. | 2.90 |

 Download This Application
Download This Application