Analysis of Cloperastine Fendizoate listed in the Japanese Pharmacopoeia 18th Edition
April 21, 2026Introduction
Cloperastine fendizoate, newly listed in the 18th edition of the Japanese Pharmacopoeia (JP18), is an antitussive agent used to relieve respiratory symptoms such as coughing by acting directly on the cough center. As with all pharmacopoeial drug substances, rigorous evaluation of purity is essential to ensure product quality, safety, and compliance with regulatory standards. The JP18 monograph defines specific criteria for system suitability and analytical performance in the purity testing of cloperastine fendizoate. Verification of these parameters is critical to confirm that the chromatographic system can provide accurate, precise, and reproducible results. In this application note, we present the results of system suitability testing for the purity analysis of cloperastine fendizoate using HPLC in accordance with JP18 requirements.
Experimental
Instruments
Pump: PU-4580
Degassing unit: DG-4580
Low pressure gradient unit: LG-4580
Autosampler: AS-4550
Column oven: CO-4060
UV detector: UV-4570
LC Conditions
Column: Unifinepak C18 (4.6 mm I.D. x 150 mm L, 5 µm)
Eluent A: 0.1 M Potassium dihydrogen phosphate /acetonitrile/perchloric acid (400/320/1)
Eluent B: Acetonitrile/0.1 M potassium dihydrogen phosphate/perchloric acid (1050/450/1)
Gradient: A/B = 100/0 (0.0 min) → 100/0 (12.0 min) → 0/100 (22.0 min) → 0/100 (27.0 min) → 100/0 (27.1 min), 1 cycle; 37 min
Flow rate: 1.2 mL/min
Column temp: 25 ºC
Wavelength: 226 nm
Injection volume: 20 µL
Standard: 4-Cholobenzophenone
(The dissolving and diluting solvent is eluent A)
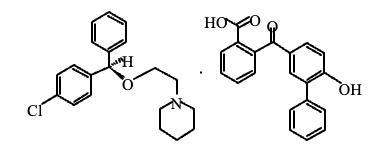
Keywords
Japanese Pharmacopoeia, cloperastine fendizoate, UV detector, system suitability
Results
An overview of the system suitability requirements for the purity analysis of cloperastine fendizoate is summarized in Table 1.
Table 1. Overview of system suitability tests for the purity analysis of cloperastine fendizoate
| Test | Criteria |
|---|---|
| Detectability | When the standard solution (1.25 mg/L) is diluted to a concentration of 0.25 mg/L and 20 µL of the diluted standard solution (0.25 mg/L) is injected under the above operating conditions, the peak area of 4-chlorobenzophenone is 14% – 26% of the peak area of a 20 µL injection of the standard solution (1.25 mg/L). |
| System Performance | When 20 µL of the standard solution (1.25 mg/L) is injected under the above operating conditions, the number of theoretical plates and the peak symmetry factor for 4-chlorobenzophenone are ≥10,000 and ≤2.0, respectively. |
| System Reproducibility | When 20 µL of the standard solution (1.25 mg/L) is injected 6 times under the above operating conditions, the relative standard deviation of the 4-chlorobenzophenone peak area is ≤2.0 %. |
Figure 2 shows the results of the purity detectability test. The ratio of the 4-chlorobenzophenone peak area for the diluted solution (0.25 mg/L) relative to the standard solution (1.25 mg/L) was 19.5%, which met the specified criterion of 14% – 26 %.
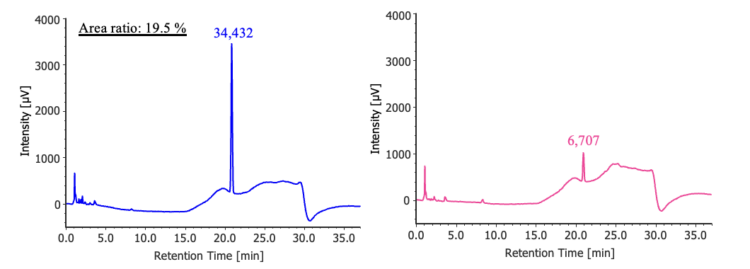
(left: 1.25 mg/L, right: 0.25 mg/L)
Figure 3 shows chromatograms of 4-chlorobenzophenone standard solutions (n = 6), and Table 2 summarizes the peak area reproducibility results based on system performance and reproducibility. The measured number of theoretical plates was 80,288 (criterion: ≥10,000), the symmetry factor was 1.01 (criterion: ≤2.0), and the relative standard deviation of the peak area was 0.21 % (criterion: ≤2.0 %). All parameters met the specified system suitability criteria.
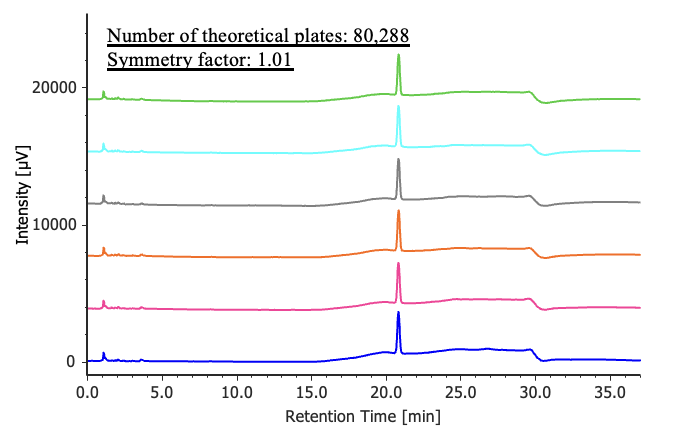
(1.25 mg/L, n = 6)
Table 2. 4-Chlorobenzophenone peak area reproducibility (n = 6)
| Injection Number | Peak Area |
|---|---|
| 1 | 33,336 |
| 2 | 33,528 |
| 3 | 33,513 |
| 4 | 33,485 |
| 5 | 33,450 |
| 6 | 33,443 |
| Average | 33,459 |
| SD | 69 |
| RSD [%] | 0.21 |
Conclusion
System suitability tests for the purity analysis of cloperastine fendizoate, which is newly listed in the 18th edition of the Japanese Pharmacopoeia (JP18), were successfully performed. As shown in Table 3, all evaluated parameters met the specified acceptance criteria of the Japanese Pharmacopoeia.
Table 3. System suitability criterion and results for the purity of cloperastine fendizoate
| Test | Test Item | Criterion | Result | Judgement |
|---|---|---|---|---|
| Detectability | Area ratio | 14–26 % | 19.5 % | Passed |
| System Performance | Number of theoretical plates | ≥10,000 | 80,288 | Passed |
| System Performance | Symmetry factor | ≤2.0 | 1.01 | Passed |
| System Reproducibility | Relative standard deviation of peak area | ≤2.0 % | 0.21 % | Passed |

 Download This Application
Download This Application