Analysis of Gefitinib listed in the Japanese Pharmacopoeia 18th Edition
April 28, 2026Introduction
Gefitinib, newly listed in the 18th edition of the Japanese Pharmacopoeia (JP18), is a therapeutic agent used in the treatment of non-small cell lung cancer (NSCLC), particularly in patients for whom surgery intervention is not feasible. It functions as a selective inhibitor of mutant epidermal growth factor receptor (EGFR) tyrosine kinase, thereby suppressing tumor cell proliferation and disease progression. In pharmaceutical quality control, accurate and reliable determination of drug purity and assay content is essential to ensure compliance with regulatory standards. The JP18 specifies analytical requirements for the evaluation of gefitinib, including system suitability criteria and method performance characteristics. This application note describes the HPLC methods for the purity and assay of gefitinib in accordance with JP18 guidelines, with a focus on system suitability evaluation, method precision, and method reproducibility to demonstrate the robustness and reliability of the analytical approach.
Experimental
Instruments
Pump: PU-4580
Degassing unit: DG-4580
Autosampler: AS-4550
Column oven: CO-4060
UV detector: UV-4570
LC Conditions
Column: Unifinepak C18 (3.0 mm I.D. x 100 mm L, 3 µm)
Eluent: Ammonium acetate solution (Dissolve 3.0 g ammonium acetate with water and fill up to 310 mL)/acetonitrile (31/19)
Flow rate: 0.9 mL/min
Column temp: 60 ºC
Wavelength: 247 nm
Injection volume: 5 µL
Standard: Gefitinib (The dissolving and diluting solvent is a mixture of 0.2 % trifluoroacetic acid aqueous solution/acetonitrile (3/2))
Keywords
Japanese Pharmacopoeia, gefitinib, UV detector, system suitability
Results
An overview of the system suitability for purity and assay tests on gefitinib is shown in Table 1.
Table 1. Overview of purity and assay system suitability tests for gefitinib
| Test | Criteria | |
|---|---|---|
| Purity | Detectability | When the purity standard solution (0.35 mg/L) is diluted to a concentration of 0.175 mg/L and the diluted purity standard (0.175 mg/L ) is injected under the above operating conditions, the signal-to-noise ratio for the gefitinib peak is ≥10. |
| Purity | System Performance | Follow the procedure for the system performance test for the assay. |
| Purity | System Reproducibility | When 5 µL of the purity standard solution is injected six times under the above operating conditions using 5 µL injections, the relative standard deviation of the gefitinib peak area is ≤2.0%. |
| Assay | System Performance | Dissolve 15 mg of 3,4-dichloroaniline in 60 mL of the assay standard solution (350 mg/L). When 5 µL of the assay standard solution (350 mg/L) is injected under the above operating conditions, 3,4-dichloroaniline and gefitinib are eluted in this order, and the resolution between the peaks is ≥5. |
| Assay | System Reproducibility | When 5 µL of the assay standard solution is injected six times under the above operating conditions, the relative standard deviation of the gefitinib peak area is ≤1.0%. |
(1) Purity
Figure 2 shows the results of the purity detectability test. The signal-to-noise ratio of the bicalutamide peak for the diluted standard solution (0.175 mg/L) was 50.7, which met the specified criterion of ≥10.
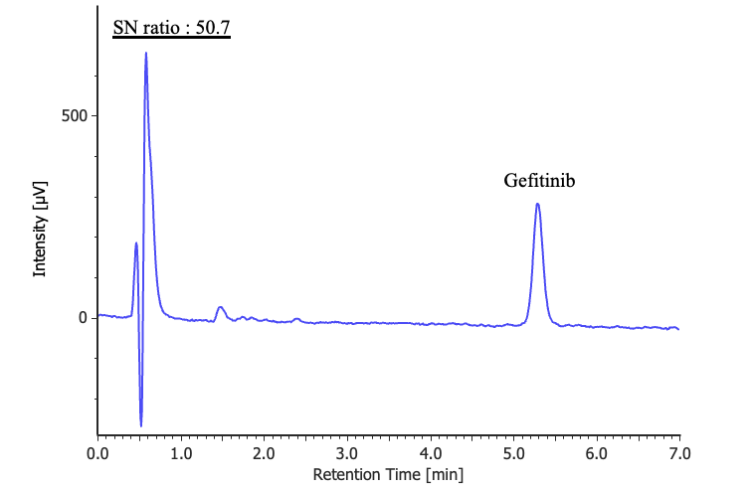
Figure 3 shows the chromatograms of the gefitinib standard solutions (n = 6), while Table 2 summarizes the peak area reproducibility results obtained from the system reproducibility test for the purity analysis. The relative standard deviation of the peak area was 0.26%, satisfying the acceptance criterion of ≤2.0%.
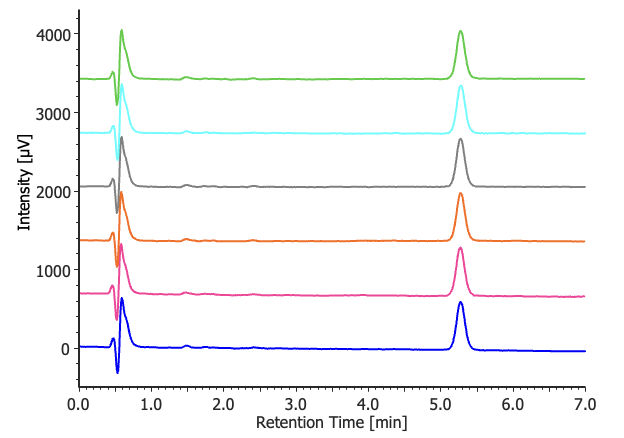
Table 2. Gefitinib peak area reproducibility (n = 6)
| Injection Number | Peak Area |
|---|---|
| 1 | 5,097 |
| 2 | 5,119 |
| 3 | 5,103 |
| 4 | 5,079 |
| 5 | 5,093 |
| 6 | 5,097 |
| Average | 5,098 |
| SD | 13 |
| RSD [%] | 0.26 |
(2) Assay
Figure 4 shows the chromatogram of a standard solution containing 3,4-dichloroaniline
(250 mg/L) and gefitinib (350 mg/L). The compounds eluted in the expected order, with
3,4-dichloroaniline preceding gefitinib. The measured resolution between the two peaks
was 9.0, satisfying the JP18 acceptance criterion of ≥5.
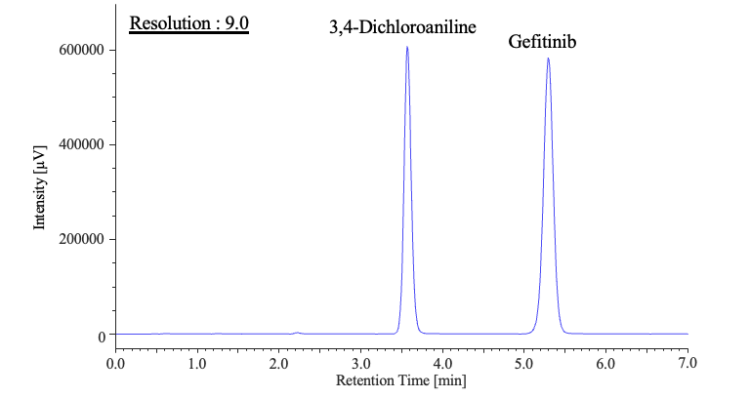
Figure 5 shows the chromatograms of the assay standard solution (n = 6), while Table 3 summarizes the corresponding system suitability results. The relative standard deviation of the gefitinib peak area was 0.11%, satisfying the acceptance criterion of ≤1.0%.
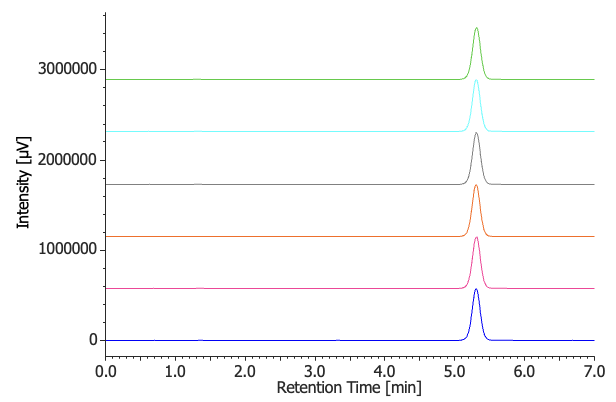
Table 3. Gefitinib peak area reproducibility (n = 6)
| Injection Number | Peak Area |
|---|---|
| 1 | 4,922,389 |
| 2 | 4,932,506 |
| 3 | 4,937,250 |
| 4 | 4,926,834 |
| 5 | 4,932,461 |
| 6 | 4,930,941 |
| Average | 4,930,397 |
| SD | 5,153 |
| RSD [%] | 0.11 |
Conclusion
System suitability testing for both the purity and assay methods of gefitinib, as specified in the 18th edition of the Japanese Pharmacopoeia, was successfully performed. As summarized in Table 4, all evaluated parameters met the specified acceptance criteria of the Japanese Pharmacopoeia.
Table 4. System suitability criterion and results for the purity and assay tests of gefitinib
| Test | Test Item | Criterion | Result | Judgement | |
|---|---|---|---|---|---|
| Purity | Detectability | Signal-to-noise ratio | ≥ 10 | 50.7 | Passed |
| Purity | System Reproducibility | Relative standard deviation of peak area | ≤ 2.0 % | 0.26 % | Passed |
| Assay | System Performance | Resolution | ≥ 5 | 9.0 | Passed |
| Assay | System Reproducibility | Relative standard deviation of peak area | ≤ 1.0 % | 0.11 % | Passed |

 Download This Application
Download This Application