Circular Dichroism Spectrum QC Test Program
August 15, 2022
Introduction

Biopharmaceutical and biotech product production is a long process that entails years of research, development, and clinical trials. These phases demand rigorous chemical stability studies for optimization of dosage and product scale up, as well as suitability of product packaging. Circular dichroism (CD) is a commonly used technique in the pharmaceutical and food science industries for structural characterization of biomolecules. Changes in spectral data could indicate a loss in stability or change in structure, corresponding to inhibition of the biomolecule’s function. Differentiating between true structural changes, or differences in spectra due to instrument noise or general sample-to-sample variation, should not be left to the user’s discretion.
Comparability software allows users to compare and quantify differences between spectra by eliminating operator bias in these similarity judgements. The JWSQC-530 Spectrum QC Test program statistically evaluates the similarity and z-score between a comparison sample spectrum and reference model. Since CD monitors the difference in the absorption of left- and right-handed circularly polarized light, the measured signal is extremely small, to account for noise in the CD spectrum weighted score calculations are performed for each wavelength using the relation between the noise standard deviation and the high tension (HT) voltage of the CD spectrometer.
This application note highlights the features of the Spectrum QC Test program.
Features
- 3 algorithms available for CD: Correlation coefficient, Euclidean distance, Manhattan distance
- Sensitive to changes in spectral shape and intensity
- 21 CFR Part 1 Compliant
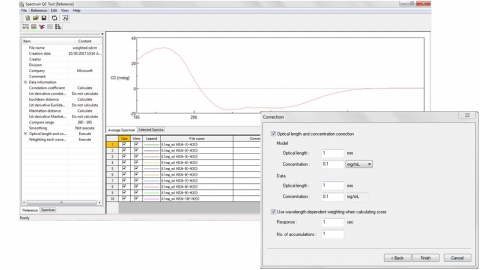
- Optical pathlength and concentration correction for reference model and sample spectrum (Figure 1)
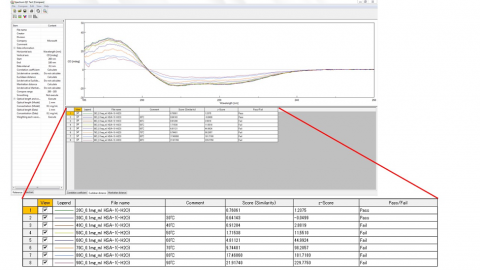
- Pass/fail evaluation function using z-score (Figure 2)
Keywords
Circular dichroism, HTCD, JWSQC-530, Pharmaceuticals, Biochemistry, Food science, Comparability
Featured Products:
-

Highest performance with a wide range of accessories for maximum flexibility to meet complex research demands.
J-1500
-
J-1700
-
High-Throughput CD

Circular Dichroism Spectrum QC Test Program
Introduction

Biopharmaceutical and biotech product production is a long process that entails years of research, development, and clinical trials. These phases demand rigorous chemical stability studies for optimization of dosage and product scale up, as well as suitability of product packaging. Circular dichroism (CD) is a commonly used technique in the pharmaceutical and food science industries for structural characterization of biomolecules. Changes in spectral data could indicate a loss in stability or change in structure, corresponding to inhibition of the biomolecule’s function. Differentiating between true structural changes, or differences in spectra due to instrument noise or general sample-to-sample variation, should not be left to the user’s discretion.
Comparability software allows users to compare and quantify differences between spectra by eliminating operator bias in these similarity judgements. The JWSQC-530 Spectrum QC Test program statistically evaluates the similarity and z-score between a comparison sample spectrum and reference model. Since CD monitors the difference in the absorption of left- and right-handed circularly polarized light, the measured signal is extremely small, to account for noise in the CD spectrum weighted score calculations are performed for each wavelength using the relation between the noise standard deviation and the high tension (HT) voltage of the CD spectrometer.
This application note highlights the features of the Spectrum QC Test program.
Features
- 3 algorithms available for CD: Correlation coefficient, Euclidean distance, Manhattan distance
- Sensitive to changes in spectral shape and intensity
- 21 CFR Part 1 Compliant
- Optical pathlength and concentration correction for reference model and sample spectrum (Figure 1)
- Pass/fail evaluation function using z-score (Figure 2)


Keywords
Circular dichroism, HTCD, JWSQC-530, Pharmaceuticals, Biochemistry, Food science, Comparability

 Download This Application
Download This Application
