Evaluation of Silica Columns for Increased Resolution in SFC separations
August 18, 2022
Introduction
SFC has been well established for enantiomeric separations and purifications in the pharmaceutical industry as well as for fuel analysis, gasoline and diesel, using the ASTM D5186 and D6550 methods. As alternative fuels are being developed, there is an increased importance on the analytical testing for comparing these new alternatives to the traditional fuels.
Both ASTM methods use a silica column for the separation, while D6550 secondarily employs a silver column for retention of the olefins after the alkanes and aromatics are separated. Our goal in this application is to further optimize the ASTM methods by evaluating a variety of silica columns to maximize the separation efficiency.

Both ASTM methods use a silica column for the separation, while D6550 secondarily employs a silver column for retention of the olefins after the alkanes and aromatics are separated. Our goal in this application is to further optimize the ASTM methods by evaluating a variety of silica columns to maximize the separation efficiency.
Experimental
| Equipment | Conditions | ||
|---|---|---|---|
| CO2 Delivery Pump: | PU-2080-CO2 | Column: | Various |
| Autosampler: | XLC-3159AS | CO2 Flow rate: | 2.0 mL/min |
| Column Oven: | CO-2060 | Column temp.: | 35oC |
| PDA Detector: | MD-2018 (High pressure cell) | Back Pressure: | Various |
| FID: | DANI Oven-FID | Wavelength: | 225 nm |
| Back Pressure Regulator: | BP-2080 | FID Splitter: | 200 oC |
| FID Detector: | 350 oC |
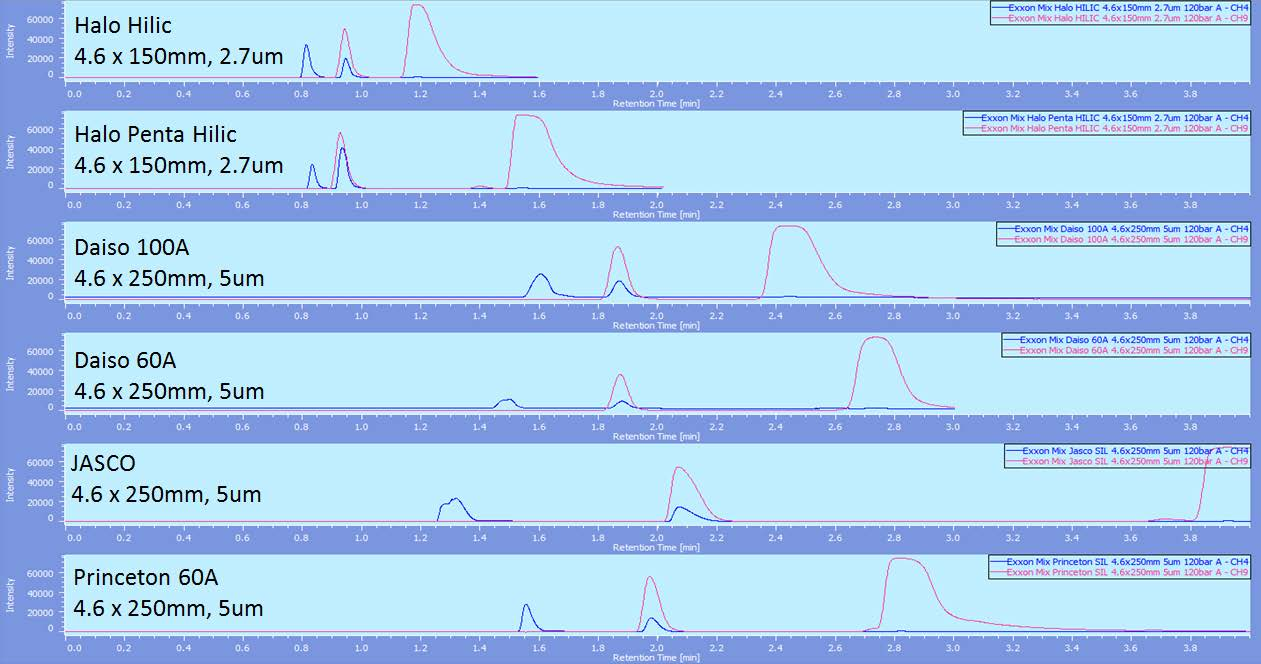
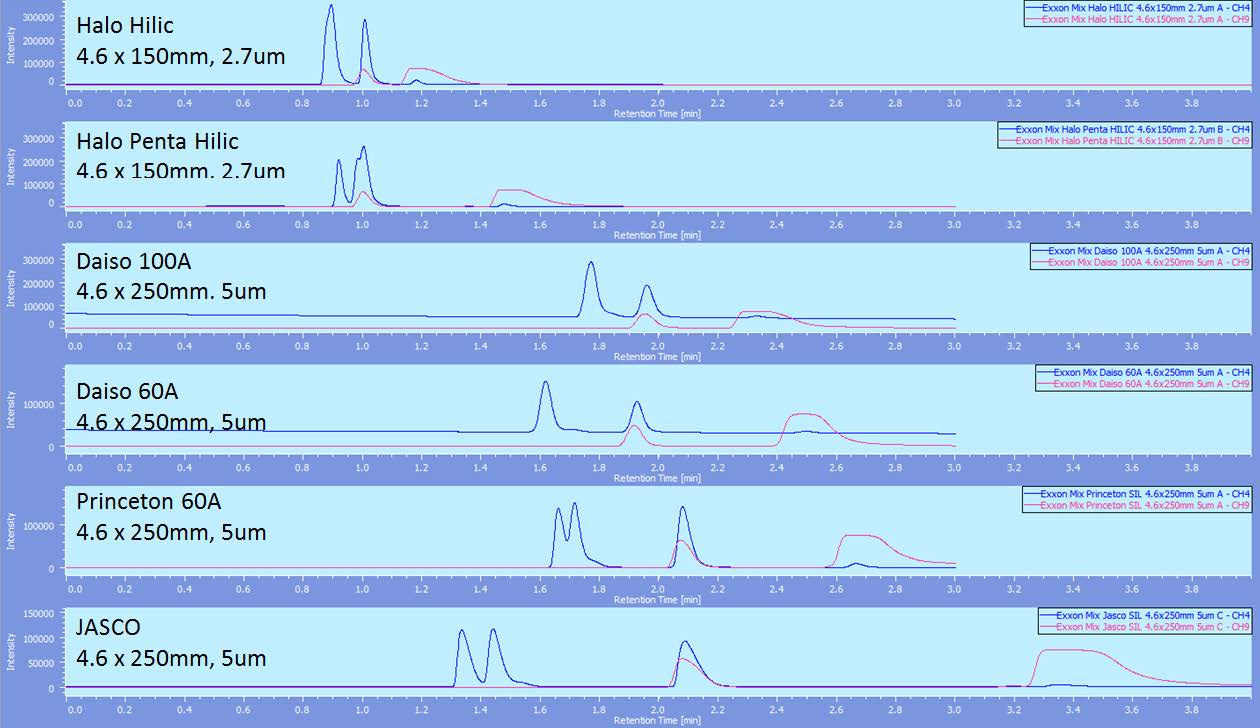
Figure 1 shows the comparison of the 6 silica columns from various manufacturers at 120 bar back pressure. The JASCO column is the only one that shows any separation of hexadecane and cyclohexane and clearly shows the most resolution between those peaks and the toluene and naphthalene peaks. Figure 2 shows this same comparison at 200 bar and again the separation of hexadecane and cyclohexane is the best and the resolution between those peaks and toluene and naphthalene is the largest.
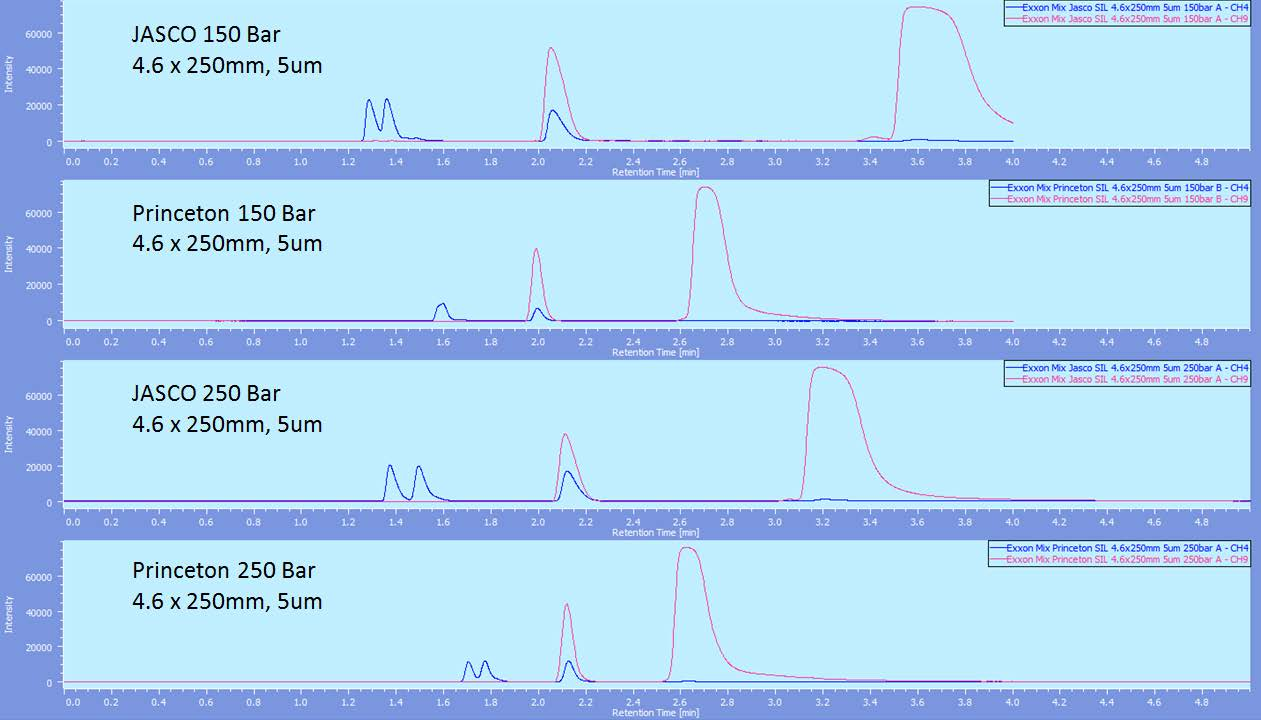
As the Princeton 60A and the JASCO columns were the only ones to show separation of hexadecane and cyclohexane, those were further evaluated at a few other pressures. As the resolution of those peaks increased when running at 200 bar compared to 120bar, 150bar and 250bar were run to confirm the trend and also that higher pressure led to the best separation.
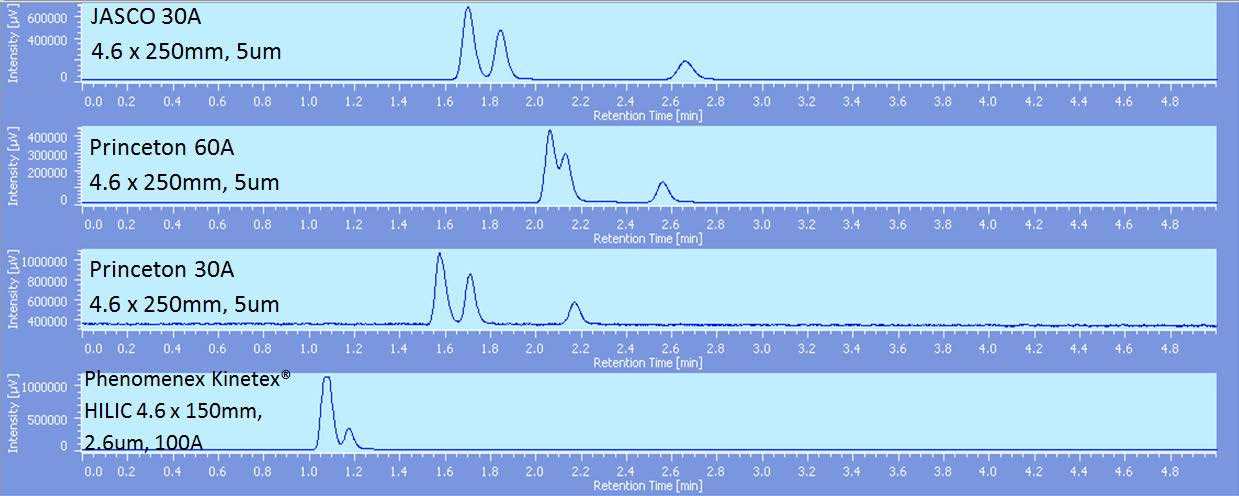
As illustrated in figure 3, an increase in pressure led to better resolution and 250bar was proven to be the best back pressure on both columns, with the JASCO column proving to be the best. As the pore size of the JASCO column is 30A, a Princeton 30A was compared as well as a Phenomenex Kinetex HILIC column using a simplified 3 component mix. As shown in figure 4 the difference between the Princeton 30A and Princeton 60A is significant as the separation of hexadecane and cyclohexane is drastically improved. The Kinetex® column did not show much retention of any of the peaks. As the pore size was 100A that was likely a factor in the insufficient resolution of hexadecane and cyclohexane.
Conclusion
The pore size is a significant factor in the separation efficiency of the column for the separation alkanes and aromatics. The JASCO 30A provided the best resolution of hexadecane, cyclohexane and toluene with the Princeton 30A a very close 2nd providing equal resolution of hexadecane and cyclohexane, but less resolution of 5.4 compared to 7.7 on the Jasco silica.
Featured Products:

Evaluation of Silica Columns for Increased Resolution in SFC separations
Introduction
SFC has been well established for enantiomeric separations and purifications in the pharmaceutical industry as well as for fuel analysis, gasoline and diesel, using the ASTM D5186 and D6550 methods. As alternative fuels are being developed, there is an increased importance on the analytical testing for comparing these new alternatives to the traditional fuels.
Both ASTM methods use a silica column for the separation, while D6550 secondarily employs a silver column for retention of the olefins after the alkanes and aromatics are separated. Our goal in this application is to further optimize the ASTM methods by evaluating a variety of silica columns to maximize the separation efficiency.

Both ASTM methods use a silica column for the separation, while D6550 secondarily employs a silver column for retention of the olefins after the alkanes and aromatics are separated. Our goal in this application is to further optimize the ASTM methods by evaluating a variety of silica columns to maximize the separation efficiency.
Experimental
| Equipment | Conditions | ||
|---|---|---|---|
| CO2 Delivery Pump: | PU-2080-CO2 | Column: | Various |
| Autosampler: | XLC-3159AS | CO2 Flow rate: | 2.0 mL/min |
| Column Oven: | CO-2060 | Column temp.: | 35oC |
| PDA Detector: | MD-2018 (High pressure cell) | Back Pressure: | Various |
| FID: | DANI Oven-FID | Wavelength: | 225 nm |
| Back Pressure Regulator: | BP-2080 | FID Splitter: | 200 oC |
| FID Detector: | 350 oC |

Figure 1 shows the comparison of the 6 silica columns from various manufacturers at 120 bar back pressure. The JASCO column is the only one that shows any separation of hexadecane and cyclohexane and clearly shows the most resolution between those peaks and the toluene and naphthalene peaks. Figure 2 shows this same comparison at 200 bar and again the separation of hexadecane and cyclohexane is the best and the resolution between those peaks and toluene and naphthalene is the largest.

As the Princeton 60A and the JASCO columns were the only ones to show separation of hexadecane and cyclohexane, those were further evaluated at a few other pressures. As the resolution of those peaks increased when running at 200 bar compared to 120bar, 150bar and 250bar were run to confirm the trend and also that higher pressure led to the best separation.

As illustrated in figure 3, an increase in pressure led to better resolution and 250bar was proven to be the best back pressure on both columns, with the JASCO column proving to be the best. As the pore size of the JASCO column is 30A, a Princeton 30A was compared as well as a Phenomenex Kinetex HILIC column using a simplified 3 component mix. As shown in figure 4 the difference between the Princeton 30A and Princeton 60A is significant as the separation of hexadecane and cyclohexane is drastically improved. The Kinetex® column did not show much retention of any of the peaks. As the pore size was 100A that was likely a factor in the insufficient resolution of hexadecane and cyclohexane.

Conclusion
The pore size is a significant factor in the separation efficiency of the column for the separation alkanes and aromatics. The JASCO 30A provided the best resolution of hexadecane, cyclohexane and toluene with the Princeton 30A a very close 2nd providing equal resolution of hexadecane and cyclohexane, but less resolution of 5.4 compared to 7.7 on the Jasco silica.

 Download This Application
Download This Application

