Parallel SFC: A Faster Approach to Chiral Column Screening
April 16, 2026Introduction
Supercritical fluid chromatography (SFC) is increasingly utilized for the separation and purification of optical isomers (chiral compounds) due to its advantages over high-performance liquid chromatography (HPLC), including faster analysis times, reduced consumption of organic solvents, and simplified post-processing. SFC systems enable rapid identification of optimal separation conditions through automated variation of column chemistries (column screening) and modifier solvent compositions, facilitating efficient chiral separations at the analytical scale. Once suitable conditions are established, methods can be readily scaled up for preparative purification. Since SFC is faster and more environmentally friendly compared to HPLC, it has been widely adopted in pharmaceutical drug discovery, particularly in response to increasingly stringent regulations governing the use and disposal of organic solvents
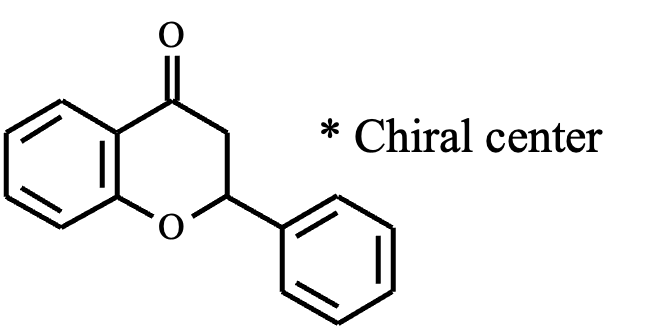
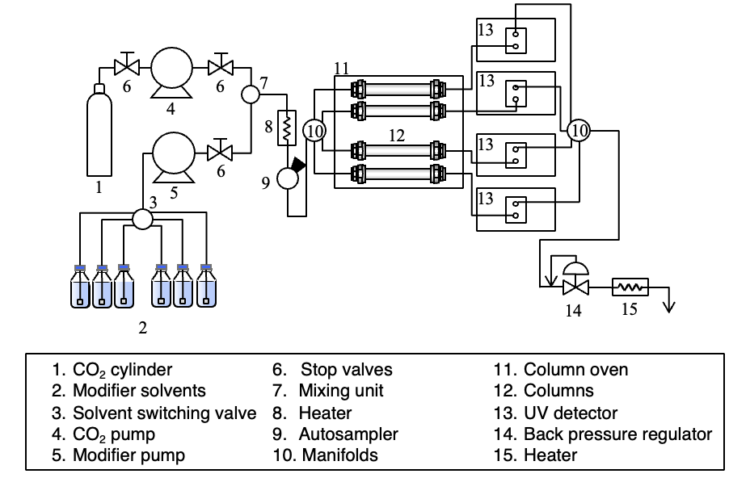
There are two primary approaches used for column screening in SFC. In the first approach, the desired column is selected by switching valves positioned before and after the column. In the second approach, a parallel screening method is employed in which the flow path is divided at a manifold, allowing the mobile phase and the sample to be delivered simultaneously to multiple columns. This parallel configuration enables concurrent analyses, significantly reducing overall screening time. In this application note, we demonstrate the separation of flavanone, a chiral compound, using the parallel SFC system.
Experimental
Instruments
CO2 pump: PU-4386
Modifier pump: PU-4086*
Heater: HE-02
Heater controller: HC-4068-01
Autosampler: AS-4350
Column oven: CO-4060*
UV dectector: UV-4075* x 4
BP regulator: BP-4340
*with option units
SFC Conditions
Column: CHIRALPAK IA, IB, IC, ID* (4.6 mm I.D. x 150 mm L, 5 µm)
Eluent A: Carbon dioxide
Eluent B: Acetonitrile, methanol
Flow rate: Eluent A; 8.0 mL/min, Eluent B; 3.0 mL/min
Column temp: 40 ºC
Wavelength: 250 nm
Back pressure: 15 MPa
Injection volume: 10 µL
Sample: 1 mg/mL flavanone in MeOH
*CHIRALPAK is a trademark or registered mark of Daicel Corporation.
Keywords
SFC, supercritical fluid chromatography, Chiral compounds, Column screening, CHIRALPAK, UV detector
Results
Figure 3 shows chromatograms of flavanone obtained using the parallel SFC system. Acetonitrile and methanol were evaluated as modifier solvents, and both achieved baseline separation within 4 minutes. The results indicate that, with both solvents, racemic flavanone can be successfully resolved on CHIRALPAK IA and CHIRALPAK ID columns.
On the CHIRALPAK IA column, shorter retention times were observed with acetonitrile compared to methanol, whereas the opposite trend was observed on the CHIRALPAK ID column. The reproducibility of retention times and peak areas for both columns and modifier solvents is summarized in Tables 1 to 4.
For comparison, performing the same analyses using the column-switching method required approximately 46 minutes to obtain four chromatograms, including column equilibration time. In contrast, the parallel method reduced total analysis time by more than an order of magnitude. Furthermore, although only two modifier solvents were evaluated in this study, expanding the number of solvents (up to ten) would further enhance the time-saving advantages of the parallel screening approach over conventional column switching.
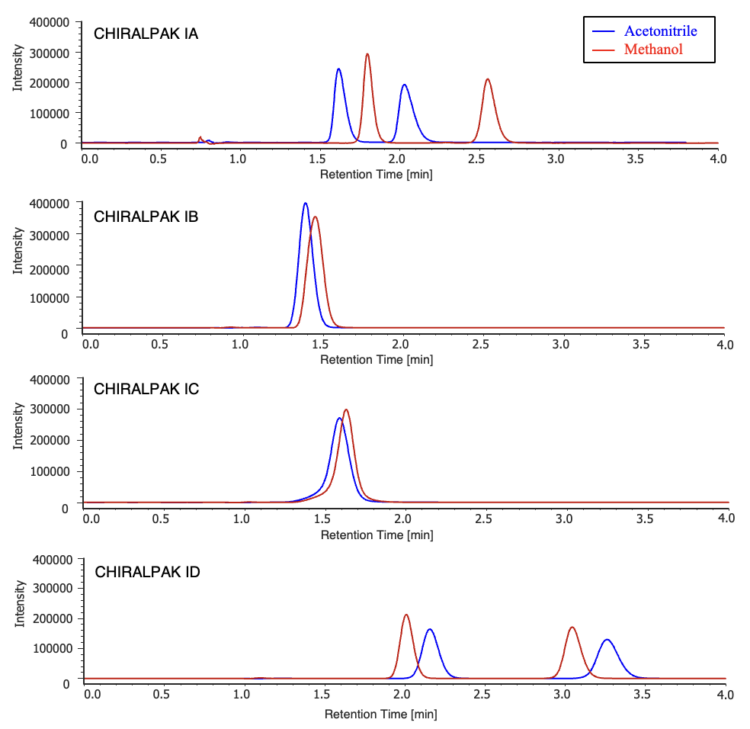
Table 1. Reproducibility of measurement results obtained using parallel SFC system (modifier solvent: acetonitrile, column: CHIRALPAK IA)
| Injection Number | Flavanone 1 | Flavanone 1 | Flavanone 2 | Flavanone 2 |
|---|---|---|---|---|
| tR [min] | Peak Area | tR [min] | Peak Area | |
| 1 | 1.627 | 1,232,807 | 2.046 | 1,232,651 |
| 2 | 1.633 | 1,258,190 | 2.045 | 1,248,113 |
| 3 | 1.617 | 1,241,883 | 2.029 | 1,240,881 |
| 4 | 1.624 | 1,241,476 | 2.039 | 1,230,797 |
| 5 | 1.627 | 1,258,181 | 2.040 | 1,246,964 |
| Average | 1.626 | 1,246,507 | 2.040 | 1,239,881 |
| SD | 0.005 | 10,071 | 0.006 | 7,124 |
| RSD [%] | 0.320 | 0.808 | 0.296 | 0.575 |
Table 2. Reproducibility of measurement results obtained using parallel SFC system (modifier solvent: acetonitrile, column: CHIRALPAK ID)
| Injection Number | Flavanone 1 | Flavanone 1 | Flavanone 2 | Flavanone 2 |
|---|---|---|---|---|
| tR [min] | Peak Area | tR [min] | Peak Area | |
| 1 | 2.177 | 1,231,384 | 3.309 | 1,231,448 |
| 2 | 2.176 | 1,255,521 | 3.288 | 1,253,962 |
| 3 | 2.157 | 1,238,788 | 3.261 | 1,241,872 |
| 4 | 2.165 | 1,241,112 | 3.288 | 1,245,737 |
| 5 | 2.163 | 1,247,816 | 3.275 | 1,254,756 |
| Average | 2.168 | 1,242,924 | 3.284 | 1,245,555 |
| SD | 0.008 | 8,200 | 0.016 | 8,578 |
| RSD [%] | 0.357 | 0.660 | 0.485 | 0.689 |
Table 3. Reproducibility of measurement results obtained using parallel SFC system (modifier solvent: methanol, column: CHIRALPAK IA)
| Injection Number | Flavanone 1 | Flavanone 1 | Flavanone 2 | Flavanone 2 |
|---|---|---|---|---|
| tR [min] | Peak Area | tR [min] | Peak Area | |
| 1 | 1.782 | 1,272,380 | 2.531 | 1,270,850 |
| 2 | 1.797 | 1,266,372 | 2.553 | 1,272,041 |
| 3 | 1.787 | 1,262,603 | 2.538 | 1,264,940 |
| 4 | 1.788 | 1,249,751 | 2.545 | 1,253,775 |
| 5 | 1.797 | 1,256,198 | 2.554 | 1,263,684 |
| Average | 1.790 | 1,261,461 | 2.544 | 1,265,058 |
| SD | 0.006 | 7,867 | 0.009 | 6,504 |
| RSD [%] | 0.330 | 0.624 | 0.346 | 0.514 |
Table 4. Reproducibility of measurement results obtained using parallel SFC system (modifier solvent: methanol, column: CHIRALPAK ID)
| Injection Number | Flavanone 1 | Flavanone 1 | Flavanone 2 | Flavanone 2 |
|---|---|---|---|---|
| tR [min] | Peak Area | tR [min] | Peak Area | |
| 1 | 1.995 | 1,240,478 | 3.026 | 1,244,109 |
| 2 | 2.010 | 1,251,413 | 3.045 | 1,253,958 |
| 3 | 2.000 | 1,238,216 | 3.028 | 1,241,664 |
| 4 | 2.000 | 1,218,774 | 3.035 | 1,234,367 |
| 5 | 2.010 | 1,236,875 | 3.042 | 1,241,633 |
| Average | 2.003 | 1,237,151 | 3.035 | 1,243,146 |
| SD | 0.006 | 10,520 | 0.007 | 6,315 |
| RSD [%] | 0.300 | 0.850 | 0.246 | 0.508 |

 Download This Application
Download This Application