Aflatoxins Analysis in Food by HPLC and UHPLC | JASCO
January 5, 2024
Introduction

Aflatoxins are a group of mycotoxins produced by microorganisms such as Aspergillus flavus, Aspergillus parasiticus and Aspergillus nomius living in tropical or subtropical regions and have a strong carcinogenic affect. It has been reported that higher levels of aflatoxins are often found which exceed the safe levels laid down by food safety agencies such as the FDA in wide variety of food products including fruits, pulses, grains and spices.
Present regulations require that total aflatoxins (sum of aflatoxin B1, B2, G1 and G2) must be lower than 10 μg/kg. A precolumn derivatization method that uses trifluoroacetic acid (TFA) in combination with HPLC with fluorescence detection offers greatly improved sensitivity for aflatoxins B1 and G1. In addition to the derivatization method, the utilization of multi-functional columns or immunoaffinity columns can be used to improve the reproducibility and recovery in the sample preparation procedure.
Experimental
Conventional HPLC
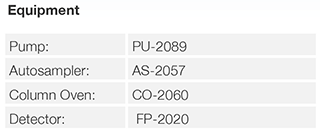
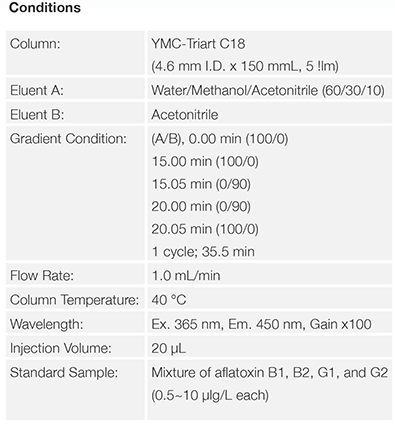
UHPLC
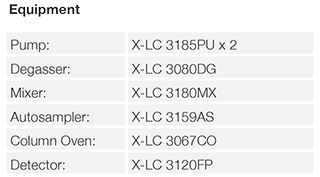
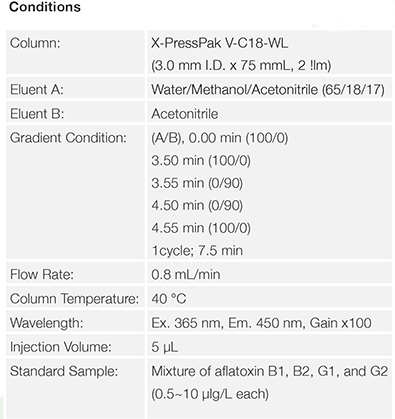
Structure
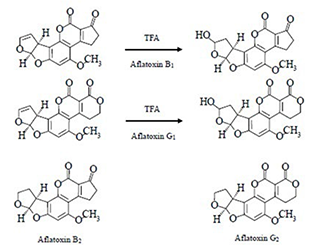
Aflatoxin B1, B2, G1 and G2 are compounds that yield native fluorescence. However, the fluorescence intensity of B1 and G1 is much less in comparison to that of B2 and G2 and accordingly, the sensitivity of B1 and G1 must be improved by changing from the natural form into a hydroxidized form using TFA derivatization. The structure of aflatoxin B1, B2, G1 and G2 and the structure of derivatized B1 and G1 are shown in figure. 1.
Derivatizaton
The procedures for TFA derivatization of the standard mixture of aflatoxins and actual sample are shown in Figure. 2.

*1 In order to avoid the adsorption of aflatoxins onto the inner wall of vessel, the vessel must be rinsed with acetonitrile and Milli-Q water, then dried.
Sample Preparation
Corn grits and roasted peanuts were selected as test samples, and a multi-functional column was used for the sample preparation of corn grits, while an immune-affinity column was used for the preparation of roasted peanuts. The sample preparation procedures are shown in Fig. 3.
1. Corn Grits

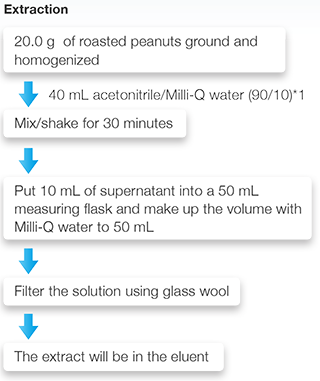
2. Roasted Peanuts
*2 Three separation modes (reverse phase, normal phase and ion-exchange) are available in the multi-functional column.(RomerLabs)
*3 Immunoaffinity column (HORIBA) utilizing unique binding capability between anti-aflatoxin monoclonal anti-body and aflatoxins.
*4 Let the column equilibrate to room temperature. Punch a hole on the upper hole on the upper cap carefully so that the air would not pass into the gel and then take the upper and the lower cap.
*5 Make up the Milli-Q water solution with 0.20 g potassium chloride, 0.20 g potassium dihydrogen phosphate, 1.16 g of disodium hydrogen phosphate and 8.0 g of sodium chloride (pH 7.4) to 1L.
*6 Flush the column carefully so that the air will not pass into the gel.
*7 Minimize the water content of the final eluent in the acetonitrile. If the water content is relatively high in the final elution, it will take a longer for solvent elimination, which may cause denaturation of aflatoxins.
*8 To release the interacted aflatoxins from gel completely.

Keywords
630005HX
Results
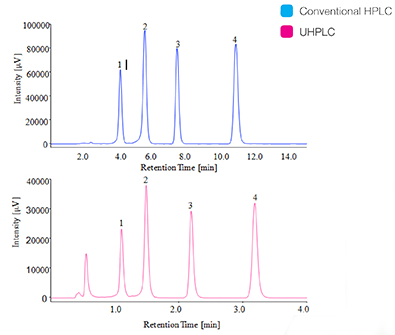
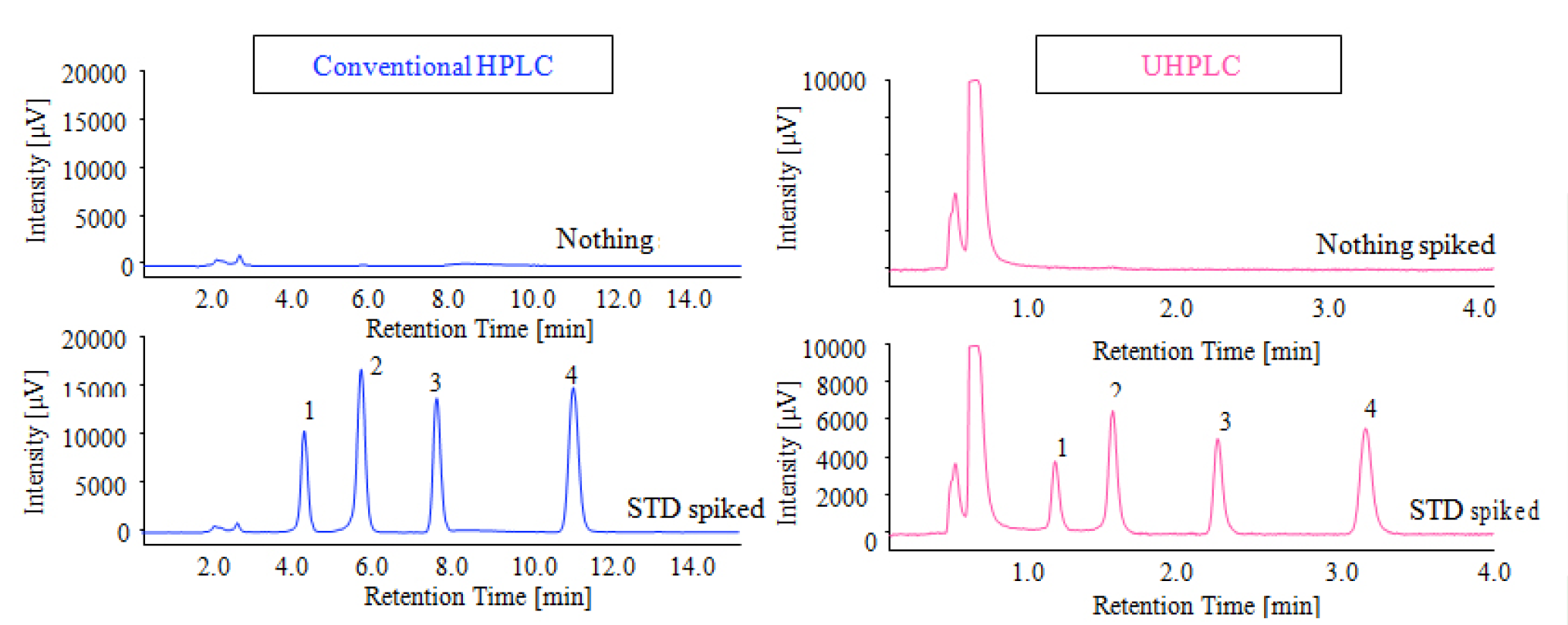
The chromatograms of standard mixtures of TFA-derivatized aflatoxins (each of 5.0μg/L) are shown in the figure 4 [by conventional HPLC (upper), by UHPLC (lower)]. The separation is completed within 12 minutes by conventional HPLC and within the 3.5 minutes by the UHPLC.
The linearity of the standard mixtures of aflatoxins is in the range 0.5 to 10μg/L with excellent co rrelation achieved r2 for both conventional HPLC and UHPLC.
Good reproducibility including the TFA derivatization procedure (N = 6) was obtained for the conventional HPLC of better than 0.2% RSD and 3% RSD in peak r etention time and area, respectively. The UHPLC also yielded good reproducibility with better than 0.2% RSD and 3.5% RSD in peak r etention time and area, respectively. The 1.0ug/L of standard mixture of aflatoxins was used for this estimation.
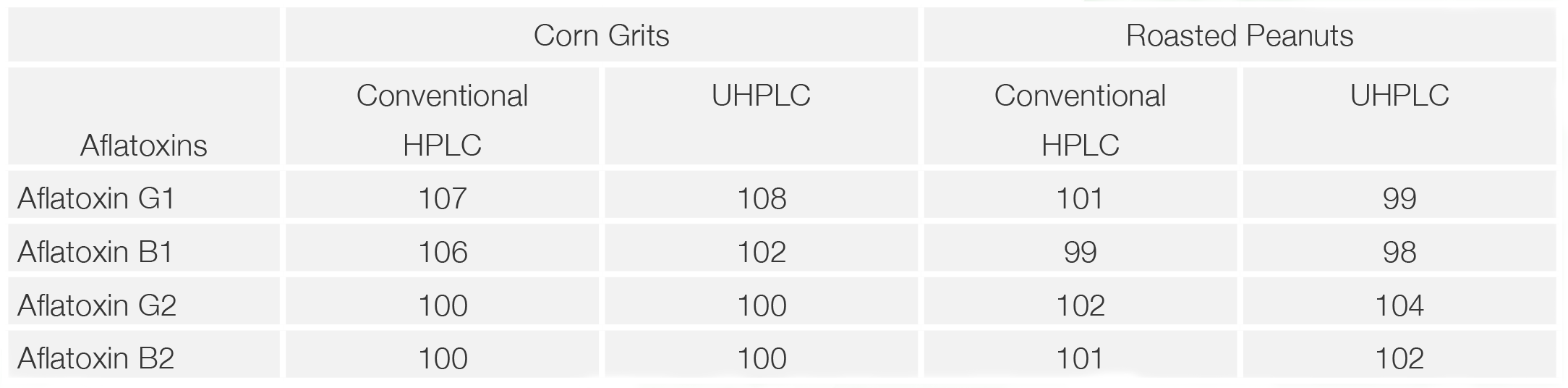
Chromatograms of corn grits samples prepared using the multi-functional columns (MycoSep 226AflaZon+) in the sample preparation are shown in figure 5 (both the conventional HPLC and UHPLC). The samples from corn grits and those spiked with a standard mixture of aflatoxins were used for the recovery estimation of aflatoxins. Almost no contaminant peaks are observed in the chromatograms and good recovery of standard aflatoxins was obtained for both conventional HPLC and UHPLC a s shown in Table 1.
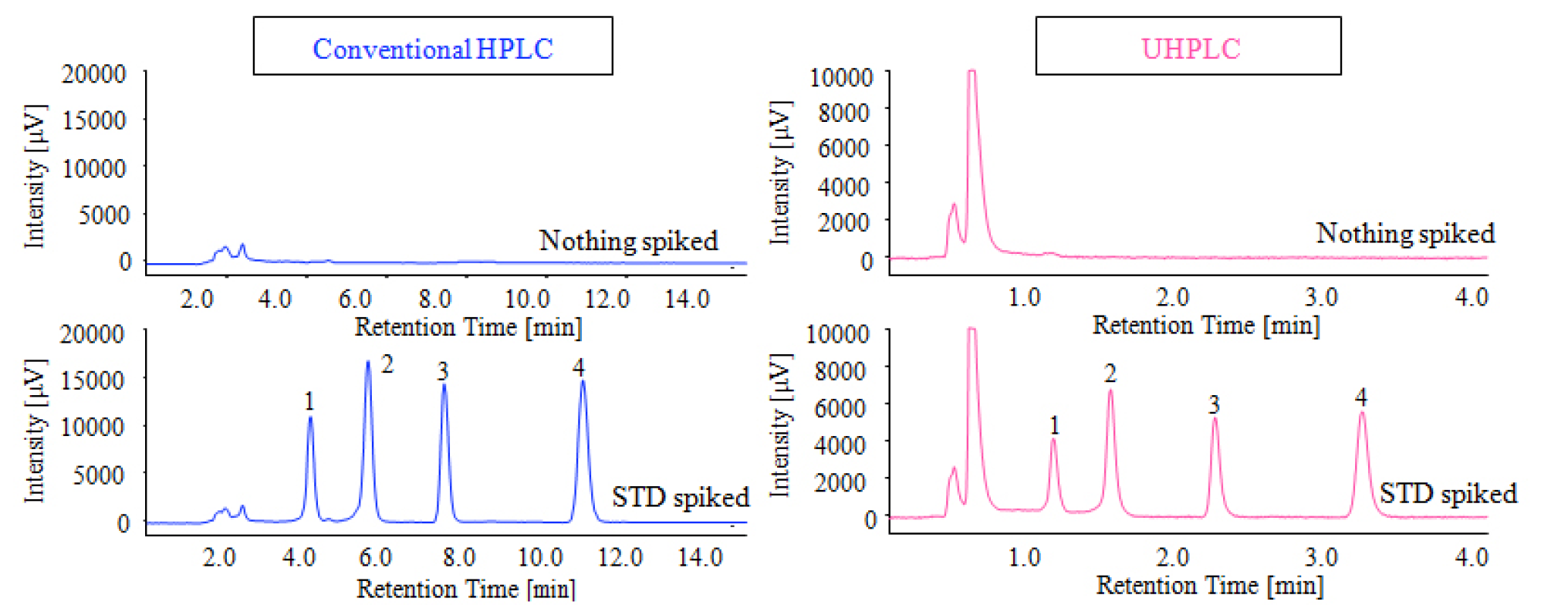
Chromatograms of the roasted peanuts sample prepared using the immunoaffinity column (AFLAKING) in sample preparation are shown in figure 6 (both the conventional HPLC and UHPLC). The samples from roasted peanuts and those spiked with the standard mixture of aflatoxins were used for the recovery estimation of aflatoxins. Almost no contaminant peaks are observed in the chromatograms and good recovery of standard aflatoxin was obtained for both conventional HPLC and UHPLC as shown in Table 2.
Featured Products:

Aflatoxins Analysis in Food by HPLC and UHPLC | JASCO
Introduction

Aflatoxins are a group of mycotoxins produced by microorganisms such as Aspergillus flavus, Aspergillus parasiticus and Aspergillus nomius living in tropical or subtropical regions and have a strong carcinogenic affect. It has been reported that higher levels of aflatoxins are often found which exceed the safe levels laid down by food safety agencies such as the FDA in wide variety of food products including fruits, pulses, grains and spices.
Present regulations require that total aflatoxins (sum of aflatoxin B1, B2, G1 and G2) must be lower than 10 μg/kg. A precolumn derivatization method that uses trifluoroacetic acid (TFA) in combination with HPLC with fluorescence detection offers greatly improved sensitivity for aflatoxins B1 and G1. In addition to the derivatization method, the utilization of multi-functional columns or immunoaffinity columns can be used to improve the reproducibility and recovery in the sample preparation procedure.
Experimental
Conventional HPLC


UHPLC


Structure
Aflatoxin B1, B2, G1 and G2 are compounds that yield native fluorescence. However, the fluorescence intensity of B1 and G1 is much less in comparison to that of B2 and G2 and accordingly, the sensitivity of B1 and G1 must be improved by changing from the natural form into a hydroxidized form using TFA derivatization. The structure of aflatoxin B1, B2, G1 and G2 and the structure of derivatized B1 and G1 are shown in figure. 1.

Derivatizaton
The procedures for TFA derivatization of the standard mixture of aflatoxins and actual sample are shown in Figure. 2.

*1 In order to avoid the adsorption of aflatoxins onto the inner wall of vessel, the vessel must be rinsed with acetonitrile and Milli-Q water, then dried.
Sample Preparation
Corn grits and roasted peanuts were selected as test samples, and a multi-functional column was used for the sample preparation of corn grits, while an immune-affinity column was used for the preparation of roasted peanuts. The sample preparation procedures are shown in Fig. 3.
1. Corn Grits

2. Roasted Peanuts

*2 Three separation modes (reverse phase, normal phase and ion-exchange) are available in the multi-functional column.(RomerLabs)
*3 Immunoaffinity column (HORIBA) utilizing unique binding capability between anti-aflatoxin monoclonal anti-body and aflatoxins.
*4 Let the column equilibrate to room temperature. Punch a hole on the upper hole on the upper cap carefully so that the air would not pass into the gel and then take the upper and the lower cap.
*5 Make up the Milli-Q water solution with 0.20 g potassium chloride, 0.20 g potassium dihydrogen phosphate, 1.16 g of disodium hydrogen phosphate and 8.0 g of sodium chloride (pH 7.4) to 1L.
*6 Flush the column carefully so that the air will not pass into the gel.
*7 Minimize the water content of the final eluent in the acetonitrile. If the water content is relatively high in the final elution, it will take a longer for solvent elimination, which may cause denaturation of aflatoxins.
*8 To release the interacted aflatoxins from gel completely.

Results
The chromatograms of standard mixtures of TFA-derivatized aflatoxins (each of 5.0μg/L) are shown in the figure 4 [by conventional HPLC (upper), by UHPLC (lower)]. The separation is completed within 12 minutes by conventional HPLC and within the 3.5 minutes by the UHPLC.
The linearity of the standard mixtures of aflatoxins is in the range 0.5 to 10μg/L with excellent co rrelation achieved r2 for both conventional HPLC and UHPLC.
Good reproducibility including the TFA derivatization procedure (N = 6) was obtained for the conventional HPLC of better than 0.2% RSD and 3% RSD in peak r etention time and area, respectively. The UHPLC also yielded good reproducibility with better than 0.2% RSD and 3.5% RSD in peak r etention time and area, respectively. The 1.0ug/L of standard mixture of aflatoxins was used for this estimation.

Chromatograms of corn grits samples prepared using the multi-functional columns (MycoSep 226AflaZon+) in the sample preparation are shown in figure 5 (both the conventional HPLC and UHPLC). The samples from corn grits and those spiked with a standard mixture of aflatoxins were used for the recovery estimation of aflatoxins. Almost no contaminant peaks are observed in the chromatograms and good recovery of standard aflatoxins was obtained for both conventional HPLC and UHPLC a s shown in Table 1.
Chromatograms of the roasted peanuts sample prepared using the immunoaffinity column (AFLAKING) in sample preparation are shown in figure 6 (both the conventional HPLC and UHPLC). The samples from roasted peanuts and those spiked with the standard mixture of aflatoxins were used for the recovery estimation of aflatoxins. Almost no contaminant peaks are observed in the chromatograms and good recovery of standard aflatoxin was obtained for both conventional HPLC and UHPLC as shown in Table 2.



Keywords
630005HX

 Download This Application
Download This Application

