High Speed Separation of Components in Cold Medicine using UHPLC
January 5, 2024
Introduction
The main components of a common cold medicine includes acetoaminophen, anhydrous caffeine, hesperidine, ethenzamide, tipepidinehibenzate, andapronalid. We examined the applicability of an X-Press Pak C18S column (2.1mm .I.D. x 50mmL.) packed with 2µm diameter packing material for the ultra-high speed separation of the above medicines. The results were examined to determine whether the performance of the column and chromatography separation exceeds that of conventional HPLC.

Experimental
The X-LC system utilized in this experiment wa sa JASCOX-LC system consisting of a 3185PU pump, 3080DG degasser, 3067CO column oven, 3070UV UV/Vis detector, 3059AS auto sampler and a chromatography data system.
Keywords
745023X
Results
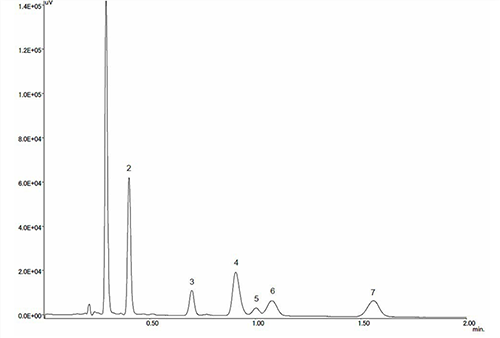
Figure 1 shows the separation of a standard mixture including acetoaminophen (0.02mg / mL), anhydrouscaffeine (0.02mg / mL), phenol (for internal standard, 0.02mg/mL), hesperidine (0.02mg/mL), ethenzamide (0.02mg/mL), tipepidinehibenzate(0.02 mg/mL) andapronalid (0.1mg/mL).TheX-LC system provides an analysis time 6 times faster than conventional HPLC without sacrificing the resolution between each peak.
Featured Products:

High Speed Separation of Components in Cold Medicine using UHPLC
Introduction
The main components of a common cold medicine includes acetoaminophen, anhydrous caffeine, hesperidine, ethenzamide, tipepidinehibenzate, andapronalid. We examined the applicability of an X-Press Pak C18S column (2.1mm .I.D. x 50mmL.) packed with 2µm diameter packing material for the ultra-high speed separation of the above medicines. The results were examined to determine whether the performance of the column and chromatography separation exceeds that of conventional HPLC.

Experimental
The X-LC system utilized in this experiment wa sa JASCOX-LC system consisting of a 3185PU pump, 3080DG degasser, 3067CO column oven, 3070UV UV/Vis detector, 3059AS auto sampler and a chromatography data system.
Results
Figure 1 shows the separation of a standard mixture including acetoaminophen (0.02mg / mL), anhydrouscaffeine (0.02mg / mL), phenol (for internal standard, 0.02mg/mL), hesperidine (0.02mg/mL), ethenzamide (0.02mg/mL), tipepidinehibenzate(0.02 mg/mL) andapronalid (0.1mg/mL).TheX-LC system provides an analysis time 6 times faster than conventional HPLC without sacrificing the resolution between each peak.

Keywords
745023X

 Download This Application
Download This Application

