HPLC of Fatty Acid Methyl Ester (FAME) and Triglyceride in Biodiesel and Diesel
January 5, 2024
Introduction
Biodiesel is made from vegetable oil produced from various plant materials into fatty acid methyl esters (FAME) by trans-esterification of the triglycerides contained in the vegetable oils. This may be an excellent replacement for non-carbon neutral diesel fuel from petroleum. The combustion of diesel oil produces carbon dioxide which may be considered to be one of the causes of global warming and is certainly considered hazardous to health.
Plant oils are produced by photosynthesis in which carbon dioxide is recovered from the atmospheric. Carbon dioxide from the combustion of biodiesel is reabsorbed by plants through photosynthesis, this offset of carbon dioxide consumption by plant material makes this source of fuel ‘carbon neutral’. As a result, determination of fatty acid methyl esters and triglycerides in fuel is now of increasing interest.
In Japan, the measurement method for determination of fatty acid methyl esters and triglycerides was promulgated in Official Gazette No.78 of the Ministry of Economy, Trade and Industry, issued 22nd of March, 2007.
JASCO has determined the fatty acid methyl esters and triglycerides content in diesel fuel mixed with biodiesel in accordance with this standard method.
Experimental

An LC-2000 series HPLC system was used, comprising of a PU-2080 pump, DG-2080-53 degasser, CO-2060 column oven, AS-2057 autosampler, RI-2031 refractive index detector, and ChromNAV chromatography data system.
The stock solution was prepared by dissolving 5.0 g of methyl stearate and 0.05 g of trilinolein in diesel oil to make up a volume of 50 mL. The standard solutions were then prepared by diluting the stock solution with diesel fuel.
Keywords
220053H
Results
Figure 1 Chromatogram obtained by injecting 5 µL of the 10x diluted stock solution. Resolution between the methyl stearate (peak 2) and trilinolein (peak 3) was calculated to be 5.7, whereas the Official Gazette’s requirement for the resolution is 3.0, well exceeding the established criteria.
In order to examine the quantitative precision, 5 µL of the stock solution and the standard solutions with various concentrations were injected and the obtained peak areas were plotted against the concentrations of the standard solutions. Results showed that the coefficients of correlation for methyl stearate was r2=1.0000 in the range of 1-100 mg/mL and for trilinolein was r2=0.9999 in the range of 0.01-1 mg/mL, respectively.
Figure 2 Shows the chromatogram obtained by injecting 5 µL of a mixture of biodiesel and diesel fuel (5/95). Peak 2, corresponding to fatty acid methyl esters, was quantified to be 48.99 mg/mL for methyl stearate. No peak corresponding to triglycerides was observed. These results meet the Official Gazette’s requirements for the contents of fatty acid methyl esters and triglycerides: less than 5.0% (w/v) and less than 0.01% (w/v), respectively.
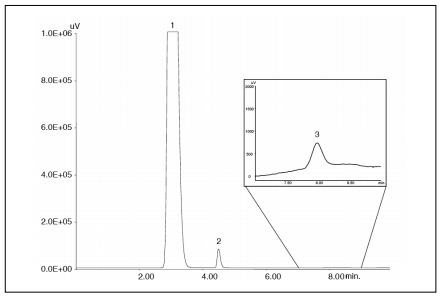
Peaks: 1 = diesel oil; 2 = methyl stearate (10 mg/mL) ; 3 = trilinolein(0.1 mg/mL)
Conditions: column = Finepak SIL-5 (4.6 mm x 250 mm); mobile phase = 2-propanol/n-hexane(0.4/99.6); flow rate = 1.0 mL/min; column temperature = 40°C; injection volume = 5 µL
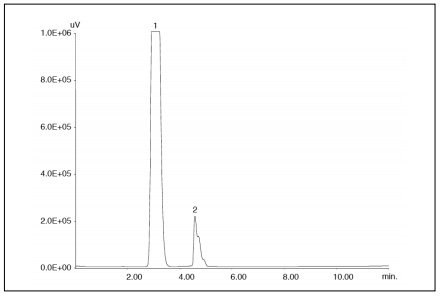
Peaks: 1 = diesel fuel; 2 = fatty acid methyl esters.
Conditions: same as in Figure 1.
Featured Products:

HPLC of Fatty Acid Methyl Ester (FAME) and Triglyceride in Biodiesel and Diesel
Introduction
Biodiesel is made from vegetable oil produced from various plant materials into fatty acid methyl esters (FAME) by trans-esterification of the triglycerides contained in the vegetable oils. This may be an excellent replacement for non-carbon neutral diesel fuel from petroleum. The combustion of diesel oil produces carbon dioxide which may be considered to be one of the causes of global warming and is certainly considered hazardous to health.
Plant oils are produced by photosynthesis in which carbon dioxide is recovered from the atmospheric. Carbon dioxide from the combustion of biodiesel is reabsorbed by plants through photosynthesis, this offset of carbon dioxide consumption by plant material makes this source of fuel ‘carbon neutral’. As a result, determination of fatty acid methyl esters and triglycerides in fuel is now of increasing interest.
In Japan, the measurement method for determination of fatty acid methyl esters and triglycerides was promulgated in Official Gazette No.78 of the Ministry of Economy, Trade and Industry, issued 22nd of March, 2007.
JASCO has determined the fatty acid methyl esters and triglycerides content in diesel fuel mixed with biodiesel in accordance with this standard method.
Experimental

An LC-2000 series HPLC system was used, comprising of a PU-2080 pump, DG-2080-53 degasser, CO-2060 column oven, AS-2057 autosampler, RI-2031 refractive index detector, and ChromNAV chromatography data system.
The stock solution was prepared by dissolving 5.0 g of methyl stearate and 0.05 g of trilinolein in diesel oil to make up a volume of 50 mL. The standard solutions were then prepared by diluting the stock solution with diesel fuel.
Keywords
220053H
Results
Figure 1 Chromatogram obtained by injecting 5 µL of the 10x diluted stock solution. Resolution between the methyl stearate (peak 2) and trilinolein (peak 3) was calculated to be 5.7, whereas the Official Gazette’s requirement for the resolution is 3.0, well exceeding the established criteria.
In order to examine the quantitative precision, 5 µL of the stock solution and the standard solutions with various concentrations were injected and the obtained peak areas were plotted against the concentrations of the standard solutions. Results showed that the coefficients of correlation for methyl stearate was r2=1.0000 in the range of 1-100 mg/mL and for trilinolein was r2=0.9999 in the range of 0.01-1 mg/mL, respectively.
Figure 2 Shows the chromatogram obtained by injecting 5 µL of a mixture of biodiesel and diesel fuel (5/95). Peak 2, corresponding to fatty acid methyl esters, was quantified to be 48.99 mg/mL for methyl stearate. No peak corresponding to triglycerides was observed. These results meet the Official Gazette’s requirements for the contents of fatty acid methyl esters and triglycerides: less than 5.0% (w/v) and less than 0.01% (w/v), respectively.

Peaks: 1 = diesel oil; 2 = methyl stearate (10 mg/mL) ; 3 = trilinolein(0.1 mg/mL)
Conditions: column = Finepak SIL-5 (4.6 mm x 250 mm); mobile phase = 2-propanol/n-hexane(0.4/99.6); flow rate = 1.0 mL/min; column temperature = 40°C; injection volume = 5 µL

Peaks: 1 = diesel fuel; 2 = fatty acid methyl esters.
Conditions: same as in Figure 1.

 Download This Application
Download This Application
