Detection of Phosphorescence from Singlet Oxygen using NIR Spectrofluorometer
January 5, 2024
Introduction

While oxygen molecules are typically in a triplet ground state, if enough energy is delivered to the molecule they can be excited to produce singlet oxygen. However, this transition is forbidden and does not proceed directly, in which case a photosensitizer is used to induce a molecular reaction. Optical irradiation excites the photosensitizer from its singlet ground state to a singlet excited state. It then changes to a triplet excited state by intersystem crossing. If the energy difference between this triplet state and the singlet ground state for the photosensitizer is almost the same as that between the singlet excited state and the triplet ground state for oxygen, energy transfer occurs. As the triplet excited photosensitizer loses its energy, singlet oxygen is produced.
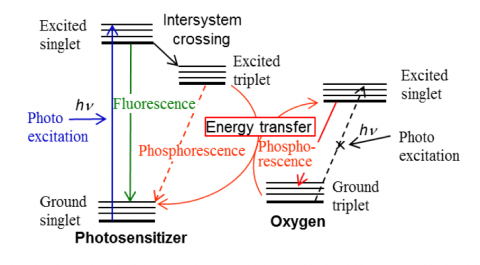
Singlet oxygen is highly active and destroys biological molecules. Recently, its use for destroying cancer cells or bacteria has been researched. Herein, we describe measurements of phosphorescence at 1270 nm emitted by singlet oxygen using a JASCO near-infrared (NIR) FP-8700 spectrofluorometer with Eosin Y as a photosensitizer.
Experimental
| Measurement Conditions | |||
| Emission Wavelength | 1200 - 1350 nm | Excitation Wavelength | 300 - 700 nm |
| Excitation Step | 5 nm | Data Interval | 2 nm |
Keywords
250-FP-0023, FP-8700, Singlet oxygen, Phosphorescence, Photosensitizer
Results
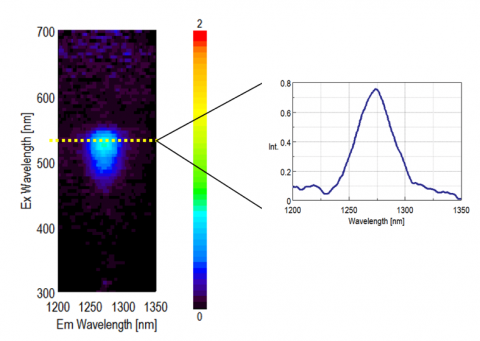
The excitation emission matrix (EEM) of EosinY ethanol solution is shown in Figure 2. The phosphorescence of singlet oxygen around 1270 nm was detected when the excitation wavelength range is 470-560 nm. The EEM measurement of singlet oxygen reveals a suitable excitation wavelength as well as whether the singlet oxygen has been generated.
Featured Products:
-

Simple and sensitive system which readily accommodates routine measurements and accessories, such as spectral scanning, quantitation, and temperature control.
FP-8250 Spectrofluorometer
-

Sophisticated optical system offering the ultimate in sensitivity, spectral accuracy, and flexibility for the most challenging materials and biological samples.
FP-8550 Spectrofluorometer
-
FP-8650 NIR Spectrofluorometer

Detection of Phosphorescence from Singlet Oxygen using NIR Spectrofluorometer
Introduction

While oxygen molecules are typically in a triplet ground state, if enough energy is delivered to the molecule they can be excited to produce singlet oxygen. However, this transition is forbidden and does not proceed directly, in which case a photosensitizer is used to induce a molecular reaction. Optical irradiation excites the photosensitizer from its singlet ground state to a singlet excited state. It then changes to a triplet excited state by intersystem crossing. If the energy difference between this triplet state and the singlet ground state for the photosensitizer is almost the same as that between the singlet excited state and the triplet ground state for oxygen, energy transfer occurs. As the triplet excited photosensitizer loses its energy, singlet oxygen is produced.

Singlet oxygen is highly active and destroys biological molecules. Recently, its use for destroying cancer cells or bacteria has been researched. Herein, we describe measurements of phosphorescence at 1270 nm emitted by singlet oxygen using a JASCO near-infrared (NIR) FP-8700 spectrofluorometer with Eosin Y as a photosensitizer.
Experimental
| Measurement Conditions | |||
| Emission Wavelength | 1200 - 1350 nm | Excitation Wavelength | 300 - 700 nm |
| Excitation Step | 5 nm | Data Interval | 2 nm |
Results

The excitation emission matrix (EEM) of EosinY ethanol solution is shown in Figure 2. The phosphorescence of singlet oxygen around 1270 nm was detected when the excitation wavelength range is 470-560 nm. The EEM measurement of singlet oxygen reveals a suitable excitation wavelength as well as whether the singlet oxygen has been generated.
Keywords
250-FP-0023, FP-8700, Singlet oxygen, Phosphorescence, Photosensitizer

 Download This Application
Download This Application