Separation Principle
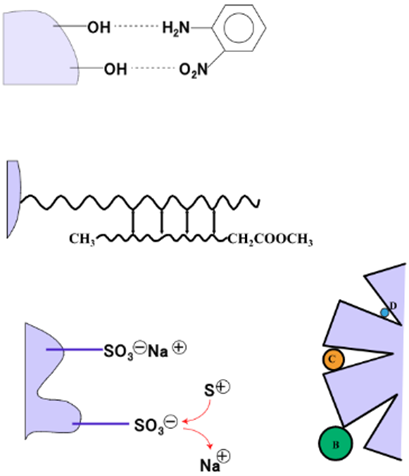
In High-Performance Liquid Chromatography (HPLC), individual components of a mixture are separated using a column based on the difference in the degree of interaction between the sample components and the stationary phase of the column. Components with a low degree of interaction with the stationary phase are eluted first. These interactions include adsorption, hydrophilic and hydrophobic interactions, electron affinity, penetration, and exclusion (Fig. 1).
Column Types and Separation Modes
As shown in the table below, there are various types of columns and separation modes that can be used, and the optimum choice depends on the nature of the sample and the analysis that is required. When an organic solvent is used as the mobile phase, a normal-phase column (mainly silica gel) can separate and analyze samples composed of fat-soluble components based on adsorption. When a water/methanol solvent composition is used as the mobile phase, a reverse-phase column (mainly carbon chains bonded to silica) can separate and analyze samples based on hydrophobic interactions. Gel Permeation Chromatography (GPC) and Gel Filtration Chromatography (GFC) columns both separate sample components based on their molecular size. The difference between the two is that GPC uses an organic solvent as a mobile phase while GFC uses an aqueous solution as the mobile phase. Finally, ion exchange columns separate and analyze samples composed of ionic components based on the electrical affinity.
| Column Type | Stationary Phase | Mobile Phase | Interaction | Features |
|---|---|---|---|---|
| Normal phase | Silica gel | Organic solvent | Adsorption | Separation of fat-soluble components |
| Reversed phase | Silica C18 (ODS) | Water / MeOH | Hydrophobic | The most common method |
| GPC (Non-aqueous) | Polymer | Organic solvent | Gel permeation | Molecular weight distribution measurement |
| GFC (Aqueous) | Hydrophilic polymer | Buffer | Gel permeation | Biopolymer separation |
| Ion exchange | Ion exchanger | Buffer | Electric affinity | Separation of ionic components |
Normal Phase vs. Reverse Phase
Normal-phase chromatography and reverse-phase chromatography are completely different separation methods. In normal-phase chromatography, a low-polarity solvent is passed through a high-polarity column, resulting in the low-polarity components being eluted first. In reverse-phase chromatography, a high-polarity solvent is passed through a non-polar column, resulting in the high-polarity components being eluted first.
Isocratic Elution vs. Gradient Elution
In HPLC, separations are commonly performed using either isocratic or gradient elution. The difference lies in how the mobile phase composition changes during the analysis. Isocratic elution occurs when the composition of the mobile phase remains constant throughout the course of the separation. Gradient elution occurs when the composition of the mobile phase changes over the course of the separation.
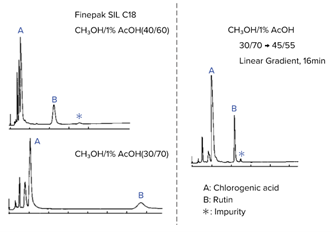
In reverse-phase chromatography and ion-exchange chromatography, gradient elution can be used to improve separation and reduce measurement time. For example, chlorogenic acid and rutin can be separated using an ODS column (Finepak SIL C18) and a methanol/1% acetic acid solution (Fig. 2).
First, let’s look at an isocratic elution of a methanol/1% acetic acid ratio of 40/60 (Fig. 2, upper left). In this example, component A could not be separated well; however, when the ratio was changed from 40/60 to 30/70 (Fig. 2, bottom left), separation was achieved, but it took a long time. By applying a gradient from a methanol/1% acetic acid ratio of 30/70 to 45/55 (changing the composition of the mobile phase over the course of the separation), the retention of the column was strengthened, resulting in the successful separation and elution of component A (Fig. 2, right).
Isocratic Elution
The mobile phase composition remains constant throughout the run.
Advantages
- Simple method development
- Stable baseline
- Reproducible retention times
- Easier troubleshooting
Limitations
- Longer run times for complex mixtures
- Late-eluting compounds may have broad peaks
- May not efficiently separate compounds with a wide range of polarities
Typical Use Cases
- Simple mixtures with similar chemical properties
- Routine quality control analyses
- Methods requiring high reproducibility and minimal complexity
Gradient Elution
The mobile phase composition changes during the run, typically increasing in solvent strength over time.
Advantages
- Faster elution of strongly retained compounds
- Sharper peaks for late-eluting analytes
- Improved separation for complex mixtures
- Better performance across a wide polarity range
Limitations
- More complex method development
- Requires precise pump mixing performance
- Baseline drift may occur due to solvent composition changes
Typical Use Cases
- Complex samples containing compounds with widely varying polarities
- Pharmaceutical impurity profiling
- Environmental or biological sample analysis
Choosing Between Isocratic and Gradient
- Use isocratic methods when analytes have similar retention characteristics and method simplicity is important.
- Use gradient methods when analyzing complex mixtures or compounds with broad retention ranges.
- Gradients are especially useful when late-eluting peaks are excessively broad or retention times are impractically long under isocratic conditions.
For practical examples of HPLC systems being used during analysis, please explore our collection of applications in the Learning Center.
FAQ
– How do I interpret a chromatogram’s baseline, peaks, retention time (tR) and dead time (t0), and when should I use peak height vs area?
A chromatogram plots detector signal (y) vs time (x). The flat line is the baseline. Each analyte appears as a peak; its apex time is the retention time (tR). The dead time (t0) is the time a non‑retained species takes to reach the detector. Peak area is generally preferred for quantitation because it’s less sensitive to peak shape; peak height can be used for narrow, well‑resolved peaks.
– What components make up an HPLC system and what does a degasser or column oven do?
Core components are the pump, degassing unit, injector/autosampler, column in a temperature‑controlled oven, detector, and data system. The degasser removes dissolved gases to prevent flow fluctuations and baseline noise/drift. The column oven stabilizes temperature to improve retention time reproducibility and resolution.
– When should I choose isocratic vs gradient elution?
Use isocratic when samples are simple and analytes have similar retention; it’s simpler and more reproducible. Use gradient when analytes span a wide polarity range or when late‑eluting peaks need to be eluted faster; gradients improve resolution and shorten run times but require gradient‑capable pumps.
– What is UHPLC and how does it differ from conventional HPLC?
UHPLC uses smaller particles (<2 µm) and higher pressures (about 600–1200 bar) than conventional HPLC (3–5 µm, up to ~600 bar). It delivers higher resolution and sensitivity, faster analyses, and reduced solvent consumption, but needs UHPLC‑rated hardware and columns.
– Which factors most affect HPLC separations?
Key drivers are analyte properties (polarity, charge, size), stationary phase chemistry, mobile phase composition and pH, flow rate, and column temperature. Selecting appropriate stationary phase and optimizing mobile phase and temperature are primary levers for resolution and selectivity.
